The mechanism of senescence – or premature cell ageing – can have an anticancer effect. This new work, conducted by Hugues de Thé and his team (Paris Diderot University/ Inserm/ CNRS/ AP-HP), was published in Nature Medicine on 12 January 2014. It reveals that targeted treatments for acute promyelocytic leukaemia, a rare form of blood cancer, cause a cascade of molecular events leading to cellular senescence and recovery. This action model could be activated in other types of cancers.
The PML/RARA* protein causes the proliferation of cancer cells in patients affected by acute promyelocytic leukaemia. Existing targeted treatments combining a hormone – retinoic acid – and a poison – arsenic – result in permanent recovery for the majority of patients, without us having a precise understanding of their action on cancer cells. Previous work by Prof Hugues de Thé’s team has shown that the combination of arsenic and retinoic acid causes destruction of the PML/RARA protein and the elimination of leukaemic stem cells. It remained to understand the link between these two events.
This new research contributes the factors needed to understand the recovery. It demonstrates the unexpected involvement of a cascade of events leading to senescence. The aim of the treatment is to reach this final ageing stage of the cells in order to render them incapable of multiplying.
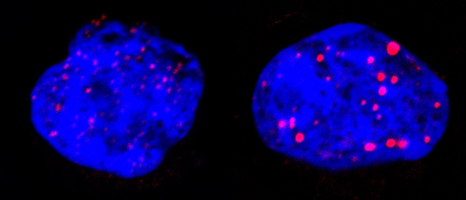
During this targeted treatment researchers showed that the p53** protein, arbiter between cell death and survival, triggers senescence through the involvement of PML nuclear bodies. These spherical structures are present in normal cells but are disorganised by PML/RARA in leukaemia. The treatment reorganises them (see figure below), activating p53 and triggering senescence. In this cascade of events (treatment, PML/RARA degradation, reformation of nuclear bodies, p53 activation), only one link has to be missing to block all the therapeutic effects.
Leukaemic cells before (left) and after treatment (right). The blue represents DNA in the nucleus; the red is nuclear corpus PML. These one are reorganised by the treatment targeting PML/RARA. © Photos provided by Prof Hugues de Thé
It is this phenomenon that enables the elimination of diseased cells and leads to total recovery of the patient, using only combined retinoic acid/arsenic treatment. The absence of chemotherapy avoids many severe side effects.
This understanding of the cellular and molecular mechanism of recovery from acute promyelocytic leukaemia opens prospects for activating this same PML/p53 pathway in other types of cancers.
This work was financed by the French Ligue contre le cancer [cancer research charity], the French Fondation ARC pour la recherche sur le cancer [ARC Foundation for Cancer Research] and the European Research Council (ERC).
* Acute promyelocytic leukaemia is caused by the modification of two genes, RAR and PML, leading to the development of cancer cells;
** the gene coding for p53 protein plays an essential role in cell proliferation under normal conditions and in maintaining the integrity of the cell genome.